Specifications
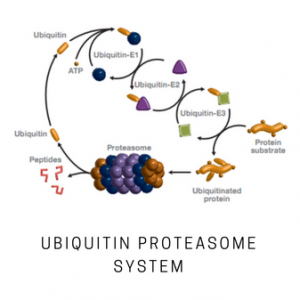
The NEDD8 activating E1 enzyme is a heterodimer composed of APPBP1 and UBA3 subunits. The APPBP1/UBA3 enzyme has homology to the N- and C-terminal halves of the ubiquitin E1 enzyme, respectively. The UBA3 subunit contains the catalytic center and activates NEDD8 in an ATP-dependent reaction by forming a high-energy thiolester intermediate. The activated NEDD8 is subsequently transferred to the UbcH12 E2 enzyme, and is then conjugated to specific substrates in the presence of the appropriate E3 ligases. This product contains a 1:1 mixture of the two subunits, forming the functional heterodimeric enzyme.
Info
| Species | Human |
| Source | E. coli |
| Tag | His6 and untagged |
| Molecular Weight | 61 kDa (APPBP1) and 54kDa (UBA3) |
| Quantity | 50 µg |
| Concentration | Variable |
| Formulation | 50 mM Tris pH 8.0, 150 mM NaCl, 10% glycerol |
| Storage | -80°C, avoid freeze/thaw cycles |


Reviews
There are no reviews yet.