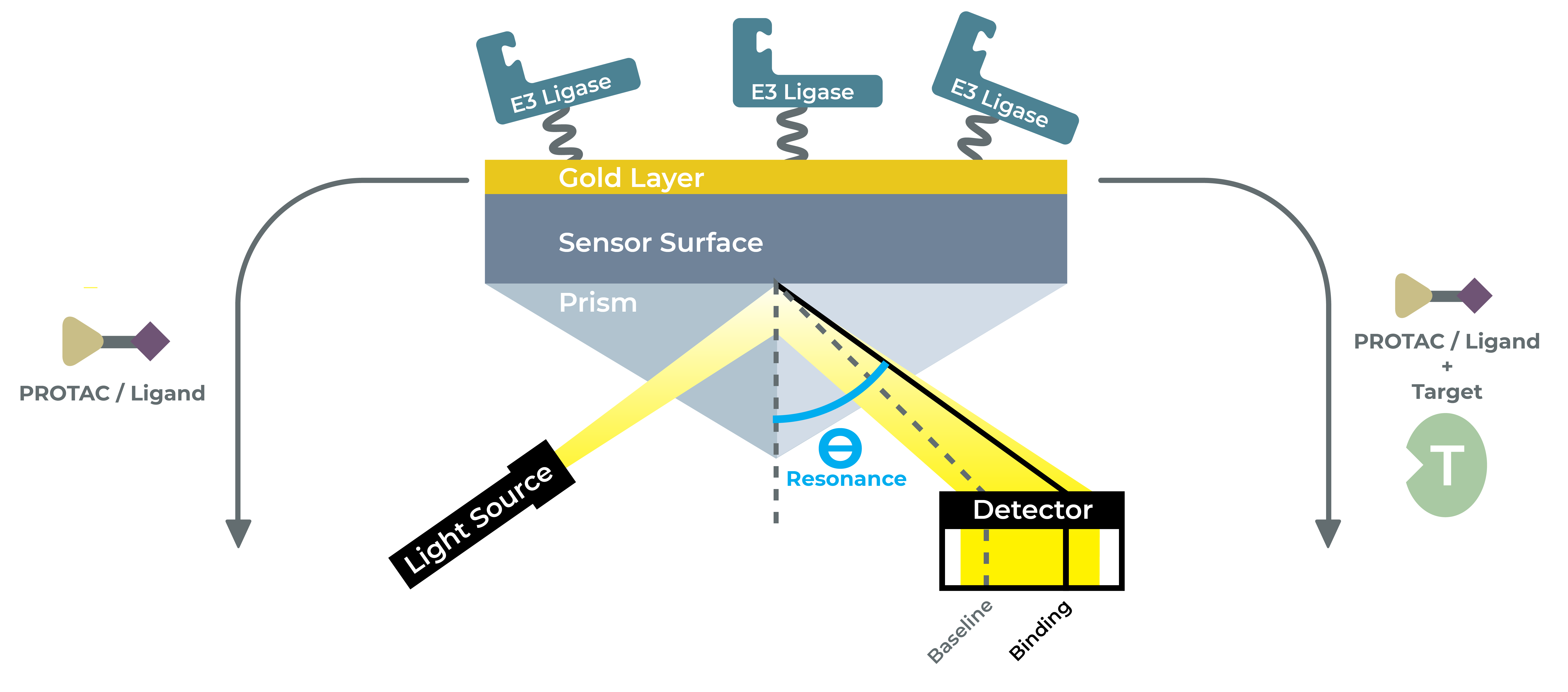
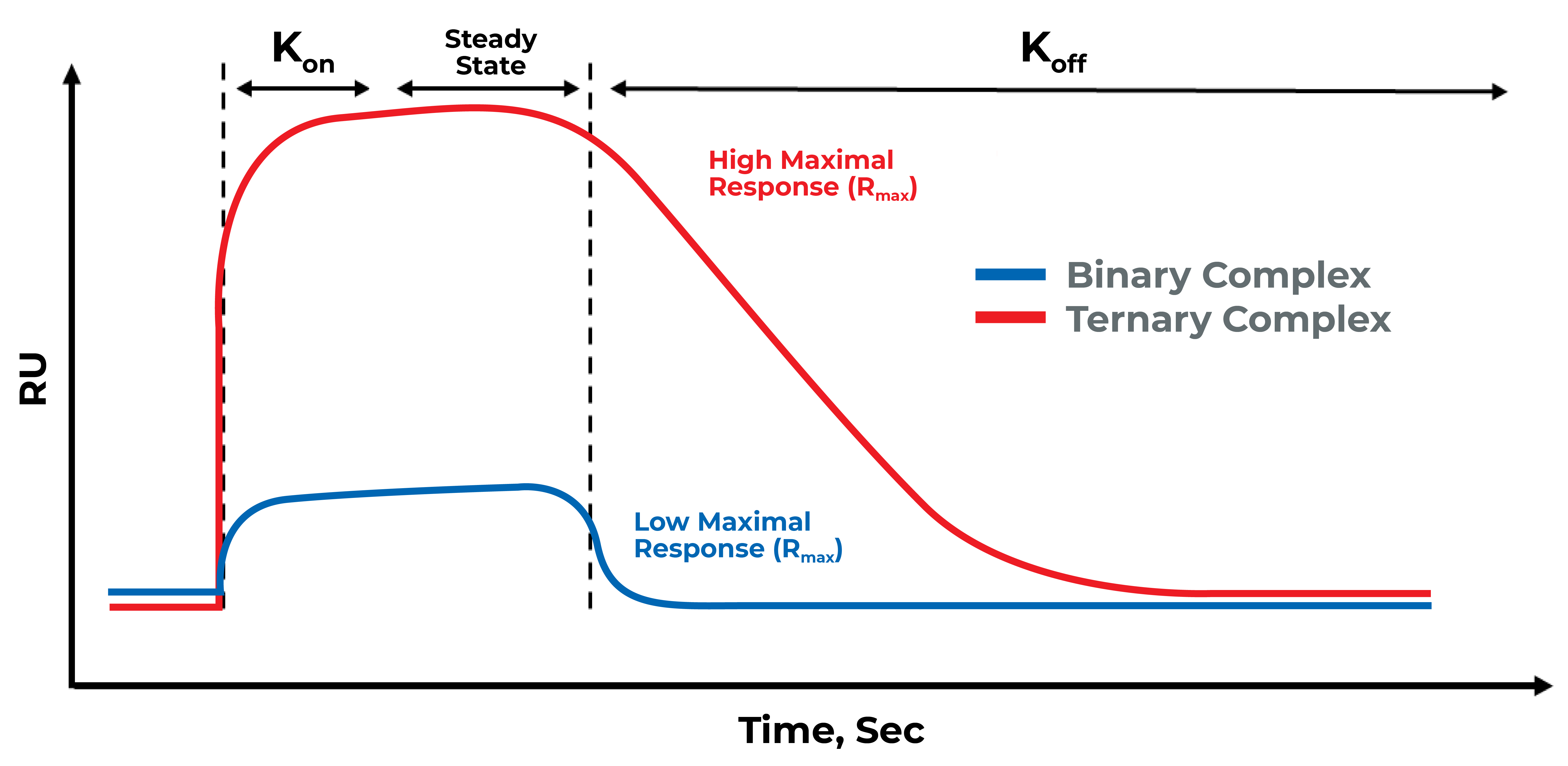
Generating experimental data for ligand binding to target protein of interest as well as determination of ligand binding affinity (Kd) is essential for drug discovery. SPR is a label-free optical technique that measures interactions in real time. A target of interest (ligand) is immobilized on a thin gold-coated glass chip while a second molecule (analyte) is flowed over the chip. Changes in the refractive index at the gold surface as the analyte binds to and dissociates from the ligand are monitored to generate sensograms, from which kon, koff, and KD can be calculated.
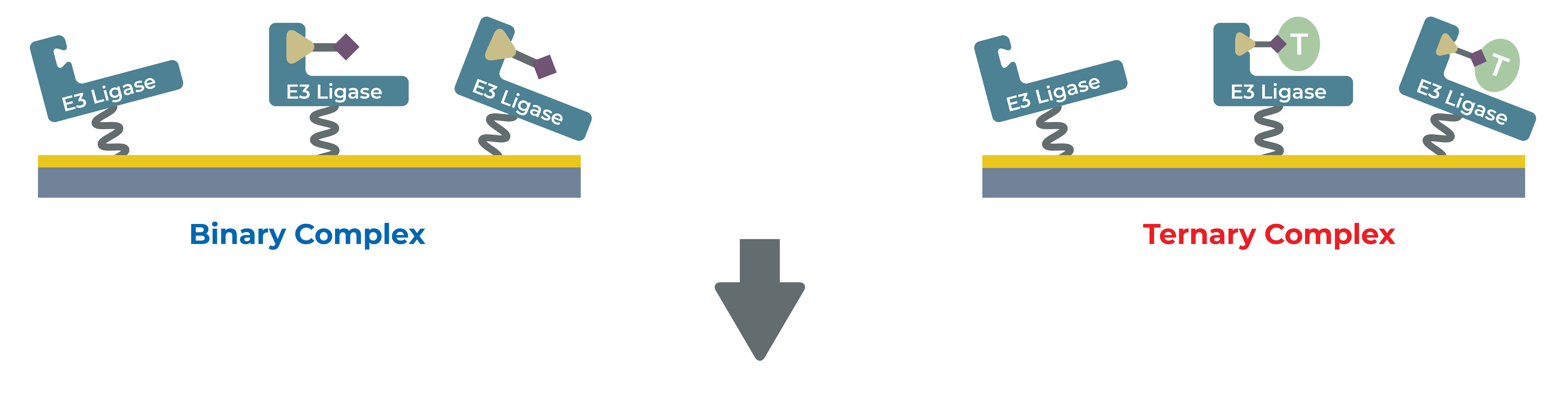
At LifeSensors we use SPR to study ligand-protein binding for drug discovery by validating hit compounds as a secondary screen. LifeSensors have established a robust protocol to study PROTACs interaction with target and E3 ligase as both binary and ternary complex formation. With over 30 ligases at our disposal, we have optimized conditions to study interaction with novel binders for PROTAC applications.
LifeSensors SPR studies are performed by highly trained technicians with thorough expertise in SPR and broad biophysics knowledge to guide clients in drug discovery of challenging targets. We use BRUKER Sierra SPR- 32 Pro high-through SPR analyzer that can enable 13,200 interactions studies (4400 samples) per day. Their breakthrough SPR+ detector allows for superior sensitivity with signal-to-noise ratio of 0.02 RU derived as a function of imaging SPR (SPRi) and high-speed optical scanning. It is crucial to have best signal-to-noise ratio when screening for identifying and characterizing novel small molecule ligands and PROTAC studies to study binary interactions.