Specifications
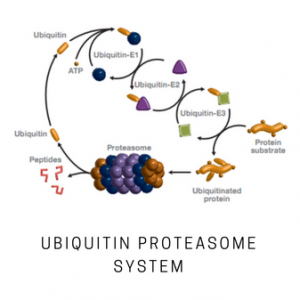
SUMO (Small Ubiquitin-related Modifier) is a ubiquitin-like family member that regulates a wide range of key cellular events. Sumoylation of proteins alters their intracellular localization, stability, and interaction with other proteins. SUMO is conjugated to its substrates utilizing a cascade of events involving activation with E1 enzyme (SAE1/SAE2), conjugation involving the E2 enzyme (UBC9), and substrate modification through the cooperation of the E2 and E3 protein ligases. SUMO and Nedd8 pathways utilize a single E1 and a single E2 in combination with a few known E3s. The dimeric activating enzyme E1 utilizes ATP to adenylate the C-terminal glycine residue of all SUMO proteins, forming a high- energy thiolester bond with the cysteine residue of SAE2.
SUMO E1 enzyme is a heterodimer of His-tagged SAE1 and un-tagged SAE2.
Info
| Species | Human |
| Source | E. coli |
| Tag | His6-tagged SAE1 and untagged SAE2 |
| Molecular Weight | 39 kDa (SAE1) and 73 kDa (SAE2) |
| Quantity | 25 µg |
| Concentration | Variable |
| Formulation | 20mM Tris, 150 mM NaCl, 2 mM βME, 10% glycerol |
| Storage | -80°C, avoid freeze/thaw cycles |


Reviews
There are no reviews yet.