DUB Screening & Profiling
Deubiquitylases or deubiquityling enzymes (DUBs) can reverse the effect of E3 ligases by removing ubiquitin from target proteins. They are also involved in ubiquitin maturation, recycling and editing. The human genome encodes ∼100 DUBs that can be divided into six subfamilies. Their substrates and the regulatory mechanisms, biological functions, and particularly the roles they play in a variety of clinical diseases, have become increasingly appreciated. Unlike most E3 ligases, DUBs have an inherent catalytic activity that is easily targeted by small molecules. At LifeSensors, we offer you products and technology that help you study DUBs.
LifeSensors has developed a variety of DUB assays utilizing physiological substrates, polyubiquitin isopeptide chain selective substrates, and several proteins attached to the C-terminus of ubiquitin that report a fluorogenic signal upon cleavage by DUBs. These substrates make up a large suite of assay technologies available to our customers. No other company provides such a wide variety of tools for HTS and physiological assays for characterizing compounds and establishing a selectivity panel. LifeSensors has expressed, purified, and characterized ~35 DUBs for HTS and compound profiling and has established DUB-centric compound libraries to provide standards for monitoring the inhibition of your compound or characterizing your DUB. LifeSensors has also developed systems for in vivo monitoring of DUB substrates.
At LifeSensors, you will find di-, tri- and tetraubiquitin chains of differing linkages for your studies and DUB-resistant polyubiquitin chains to help investigate binding interactions through pull-down studies and to serve as a control in studies of potential deubiquitylase activity towards these chains. Our lysine to arginine mutants enable studies of DUB linkage specificity and our rhodamine- or AMC- conjugated ubiquitin can be used to determine the activity and specificity of ubiquitin deconjugating enzymes (DUBs), for example, in screening.
Assays
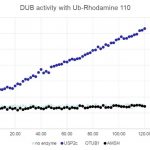
Ubiquitin Fluorophore Assay
The ubiquitin molecule is bound to a quenched fluorophore (e.g., Rhodamine 110).
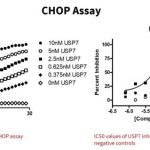
CHOP assay
The ubiquitin molecule is bound to an inactive reporter enzyme.
IQF assay
The ubiquitin molecule is bound to a fluorophore-conjugated ubiquitin molecule.
A presentation of our DUB Screening and Profiling Platform is here.