Specifications
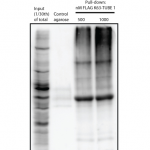
FLAG® K63 TUBE is an ideal reagent for efficient isolation and enrichment of K63-polyubiquitinated proteins from cell and tissue extracts or in vitro synthesized mixtures. FLAG® K63 TUBE consists of multiple ubiquitin interaction motifs (UIMs) joined by a rigid, helical linker that spaces the UIMs for selective binding to extended K63-linked polyubiquitin chains. The result is a peptide the exhibits high affinity binding to K63-linked polyubiquitin together with 1000 to 10,000-fold selectivity over K48- and K11- linkages. Expression of this peptide in vivo inhibits K63-linked polyubiquitin-dependent processes and protects K63-linked polyubiquitin chains from degradation. Combining this peptide with a FLAG epitope tag generates, for the first time, a powerful affinity reagent suitable for isolation, purification and characterization of proteins modified by K63-linked polyubiquitin. Flag K63-TUBE allows isolation of K63-linked polyubiquitin without the need for overexpression of ubiquitin mutants, tagged ubiquitins or the inclusion of DUB inhibitors any of which could alter cellular physiology. Isolated ubiquitinated proteins can be characterized by Western blot, mass spec or further biochemical analysis. (FLAG is a registered trademark of Sigma-Aldrich Corporation LLC).
This product is protected by one or more US or Foreign patents. Please read the Limited Use Label License to learn more. By purchasing this product, the purchaser agrees to comply with these terms.
Info
|
Tag |
FLAG |
|
Molecular Weight |
22 kDa |
|
Physical State |
Liquid |
|
Quantity |
50 µg |
|
Concentration |
Variable |
|
Storage |
-80°C, avoid freeze/thaw cycles |
This product has been cited multiple times:


Reviews
There are no reviews yet.