Specifications
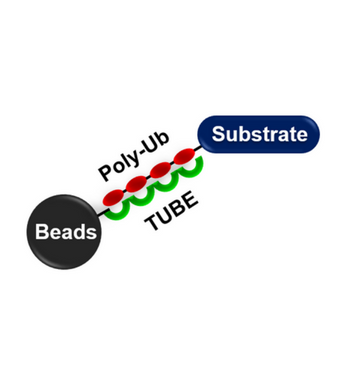
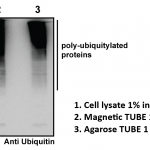
Based on protein domains known to possess an affinity for ubiquitin, Tandem Ubiquitin Binding Entities (TUBEs) have been developed for the isolation and identification of ubiquitylated proteins. TUBEs display up to a 1000-fold increase in affinity for polyubiquitin moieties over the single ubiquitin binding associated domain (UBA). In addition, TUBEs display a protective effect on polyubiquitinated proteins, allowing for detection at relatively low abundance. These properties effectively “capture” protein in its polyubiquitinated state. TUBEs can be used to isolate, enrich, and identify ubiquitinated proteins from cell and tissue extracts. Magnetic beads coupled to TUBE 1 allow for efficient recovery of polyubiquitinated proteins in a single step without centrifuge. The magnetic TUBEs allows more complete removal of the unbound supernatant, which leads to lower background. Polyubiquitinated proteins can easily be separated from TUBEs for proteomic studies or identification by Western blotting.
UM501M was designed by coating polymeric high capacity magnetic beads to allow superior enrichment of polyubiquitinated proteins along with minimizing non-specific binding to proteins in tissue and cellular lysates.
Info
| Tag | N/A |
| Molecular Weight | N/A |
| Physical State | Liquid |
| Quantity | 1 mL magnetic beads |
| Concentration | Variable |
| Storage | +4°C, Avoid storing below this temperature |

Reviews
There are no reviews yet.