Specifications
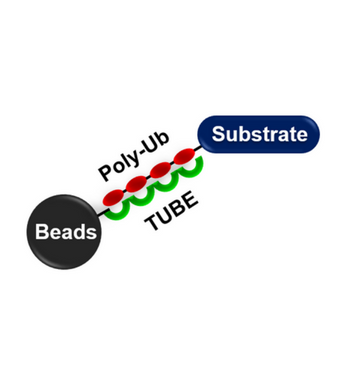
The use of Tandem Ubiquitin Binding Entities (TUBEs) is emerging as an indispensable strategy for ubiquitin research (1, 2). The first generation of these TUBEs bind K48- and K63-linked tetraUb chains with single digit nanomolar Kds, ~100 to 1000-fold more tightly than monomeric UBAs. TUBEs also protect proteins from DUBs and the proteasome, even in the absence of inhibitors normally required to block such activity. This allows efficient isolation of native polyUb chains and attached proteins from cell lines, tissues, and organs under conditions that are less likely to alter cell physiology than those listed above. TUBE 1 and TUBE 2 have recently been demonstrated to enrich for all polyUb chain linkage types, without discrimination, making these reagents appropriate even if the linkage type is not known for the protein of interest.
The K63 TUBE was developed to show enhanced selectivity for K63-linked polyubiquitin chains (~20 nM) over all other linkages (>2 µM). It can be used alone or in conjunction with our other TUBE products, especially K48 TUBE HF and M1 (linear) TUBE to investigate polyubiquitin chain linkage in your substrate protein. Magnetic-TUBEs are TUBE moieties directly coupled to magnetic beads, for the identification and characterization of polyubiquitinated proteins by western blotting and/or downstream proteomic studies. Magnetic-TUBEs facilitate the convenient “one-step” pull-down of polyubiquitinated proteins.
Info
|
Tag |
N/A |
|
Molecular Weight |
|
|
Physical State |
Liquid |
|
Quantity |
1 mL |
|
Concentration |
Variable |
|
Storage |
+4°C, avoid storing below this temperature |

Reviews
There are no reviews yet.