Specifications
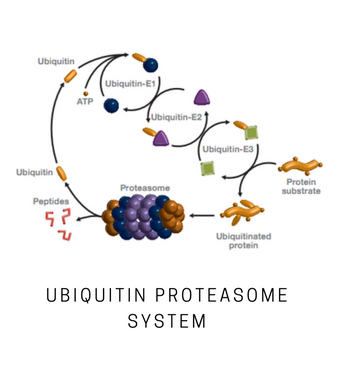
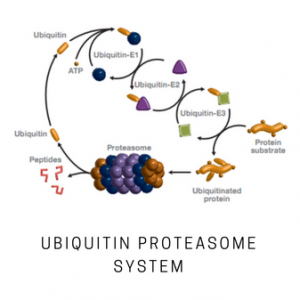
Hrd1 is a RING domain E3 ligase that is involved in the conjugation of ubiquitin to target substrates. Hrd1 has been demonstrated to function with the E2 enzymes UBE2D3 (UbcH5c) and UBE2G2 in vitro. Hrd1 is an ER-associated ligase involved in ERAD and has been linked to rheumatoid arthritis. It is present in human rheumatoid synovial cells and found to be a causative factor for arthropathy in in vivo studies. Hrd1 is also found in brain neurons and its proper regulation may be linked to neurodegenerative diseases. This construct is a Hrd1 N-terminal deletion containing residues 236-617 consisting of the complete C-terminal cytoplasmic portion. The N-terminal transmembrane spanning portion has been deleted. This Hrd1 N-terminal deletion mutant is active as an E3 ligase containing the RING and substrate recognition domains.
Hrd1 is currently made-to-order and requires a 2-3 week lead time.
Info
| Species | Human |
| Source | E. coli |
| Tag | His6-SUMO |
| Molecular Weight | Cytoplasmic fragment (236-617): 39.3 kDa (without SUMO tag) |
| Quantity | 25 μg |
| Concentration | Variable |
| Formulation | |
| Storage | -80°C, avoid freeze/thaw cycles |

Reviews
There are no reviews yet.