Specifications
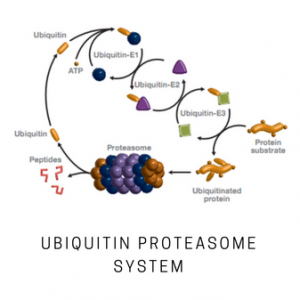
K48-linked tetraubiquitin-rhodamine provides a sensitive, high throughput means for measuring Rpn11 activity toward K48-linked chains. Cleavage of the amide bond between the C-terminal glycine of ubiquitin and rhodamine results in an increase in rhodamine fluorescence at 535 nm (Exc. 485 nm). Rpn11 (POH1), a JAMM-type isopeptidase, is one of three deubiquitinating enzymes associated with the 19S regulatory particle of the proteasome. Substrate proteins are deubiquitinated by Rpn11 prior to unfolding and translocation into the 20S for degradation. In contrast to USP14 and UCH37 (UCH-L5), isopeptidases also associated with the 19S regulatory particle, Rpn11 deubiquitination promotes substrate degradation. Therefore, modulators of Rpn11 could be effective means of controlling the function of the proteasome, a target for cancer, immune-related disorders, inflammation, neurodegeneration and other diseases. Historically, assays for Rpn11 have employed purified 26S proteasome as a source of enzyme and ubiquitinated protein as a substrate, methods requiring separation of cleaved from uncleaved substrate for determination of activity. Rpn11 activity can be distinguished from UCH37 and USP14 activity by a dependence on ATP, sensitivity to the chelator 1,10-phenanthroline (Cat. # SI9649) and insensitivity to the suicide DUB inhibitor ubiquitin-aldehyde (Cat. # SI250).
Info
| Source | E. coli |
| Tag | None |
| Molecular Weight | 34,602.7 Da (calculated) |
| Quantity | 50 µg |
| Concentration | Variable |
| Storage | -80°C, avoid freeze/thaw cycles |

Reviews
There are no reviews yet.