Specifications
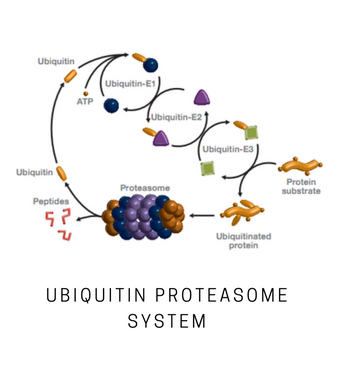
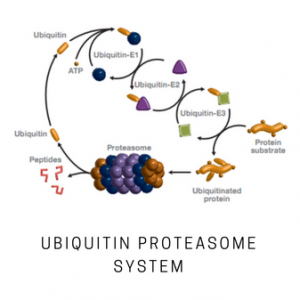
The 26S proteasome plays a central role in the ubiquitin-mediated degradation of cellular proteins. It consists of a 20S catalytic core capped by one or two 19S regulatory particles. The repeat-containing domains of ribophorins 1 and 2 (Rpn1 and Rpn2, 110 and 104 kDa, respectively) represent the largest subunits of the 26S proteasome and may also be involved in ribosome binding. Deletion of either subunit is lethal, and mutations in yeast result in impaired proteasome function and accumulation of polyubiquitinated proteins. Rpn1 is a component of the 26S proteasome base. In human, the Rpn1 subunit interacts with the ubiquitin protein ligase (E3) KIAA10. RPN1 and RPN2 form the receptors for the ubiquitin-like proteins Rad23 and Dsk2. The leucine-rich-repeat-like domain of RPN1 may participate in the recognition of the cargo proteins carried by Rad23 for unfolding and subsequent degradation. The deubiquitinating enzyme Ubp6/USP14 recognizes the proteasome base via the Rpn1 subunit. Deubiquitination by Ubp6 prevents Rpn-mediated translocation to the 20S particle.
Info
| Species | Yeast |
| Source | E. coli |
| Tag | His6 |
| Molecular Weight | 104.2 kDa |
| Quantity | 50 µg |
| Concentration | Variable |
| Formulation | 20 mM sodium phosphate pH 7.4, 500 mM NaCl, 200 mM imidazole, 1 mM β-mercaptoethanol |
| Storage | -80°C, avoid freeze/thaw cycles |

Reviews
There are no reviews yet.