Specifications
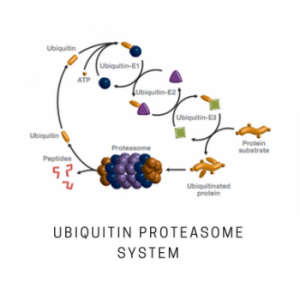
The 26S proteasome is the central protease in ubiquitin-mediated protein degradation. It is a large multi-protein complex, which plays a fundamental role in cellular homeostasis by controlling degradation of key proteins that are involved in apoptosis, cell cycle, and signal transduction. The 26S proteasome is responsible for cell quality control by eliminating misfolded proteins from the cytosol and endoplasmic reticulum. With its diverse functions, the proteasome is an important target for drug development, particularly in cancer and neurodegeneration.
A number of selective proteasome inhibitors have been identified in the last two decades and approved for clinical use (bortezomib, carfilzomib). Several other proteasomal inhibitors are currently in trials for treatment of multiple myeloma and other malignancies. The 26S proteasome is composed of a single catalytic 20S complex (core particle, CP) and one or two 19S regulatory complexes (regulatory particles, RP). The 19S regulatory complex is responsible for deubiquitylation, unfolding and re-direction of proteins to the catalytic sites within the 20S complex.
Note:
- The chymotrypsin-like activity of the 26S proteasome (45 µg/mL) was measured using 100 µM Suc-LLVY-AMC in 20 mM HEPES, pH 7.5, 0.5 mM EDTA, 0.05% Triton X, plus or minus 0.035% SDS.
- The chymotrypsin-like activity of the 26S proteasome (18 µg/mL) was measured using 100 µM Suc-LLVY-AMC in 20 mM HEPES, pH 7.5, 0.5 mM EDTA, 0.05% Triton X, plus or minus 0.5 mM MG132 (Cat. # SI9710)
Info
| Species | Human |
| Source | Human |
| Tag | N/A |
| Molecular Weight | N/A |
| Quantity | 25 µg, 50 µg |
| Concentration | 0.5 mg/mL |
| Formulation | 20 mM Tris-HCl pH 7.2, 10% Glycerol, 150 mM KCl, 1 mM β–mercaptoethanol |
| Storage | -80°C, avoid freeze/thaw cycles |


Reviews
There are no reviews yet.