By: Derrick Padykula and Isabelle Wentzel Hallmark of Neurodegeneration Diving into the complex world of neurodegeneration unveils the long road marked by the gradual decline of neuronal cells, ultimately leading to…

By: Derrick Padykula and Isabelle Wentzel Hallmark of Neurodegeneration Diving into the complex world of neurodegeneration unveils the long road marked by the gradual decline of neuronal cells, ultimately leading to…
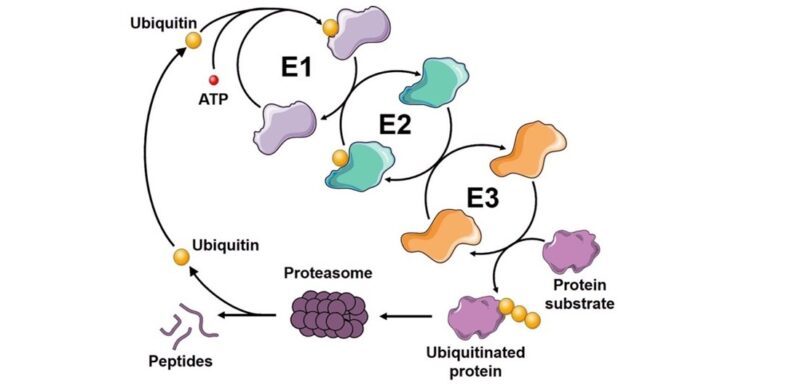
By: Derrick Padykula Ubiquitination, a ubiquitous cellular process, involves attaching ubiquitin to target proteins, influencing crucial cellular functions such as degradation, cell signaling, apoptosis, protein processing, immune response, and DNA…

MALVERN, Pa., October 26, 2023 — LifeSensors, Inc., a leading innovator in protein degradation technologies, has been invited to speak at the 6th Annual Targeted Protein Degradation Summit 2023 in…

MALVERN, Pa., SEP 21, 2023 — LifeSensors Inc., a Biotech and Drug Discovery Company focused on PROTAC, molecular glue drug discovery and neurodegeneration diagnostics has been invited to speak at the…

MALVERN, PA., May 30, 2023 — LifeSensors Inc., a Biotech and Drug Discovery Company focused on PROTACs, molecular glues, and neurodegeneration diagnostics will disclose direct methods to detect PROTAC & molecular glue…
By: Isabelle Wentzel Devastating Origins In the late 1950s and early 1960s, pregnant women who experienced nausea and morning sickness were prescribed thalidomide, a sedative drug meant to treat…
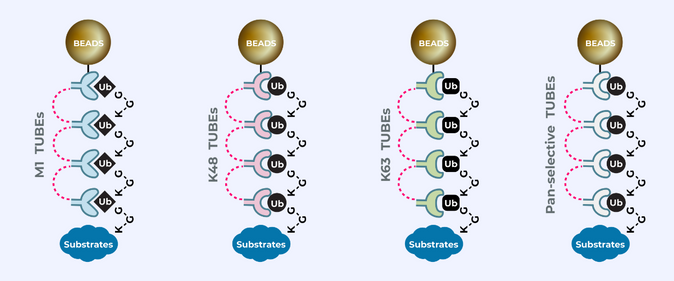
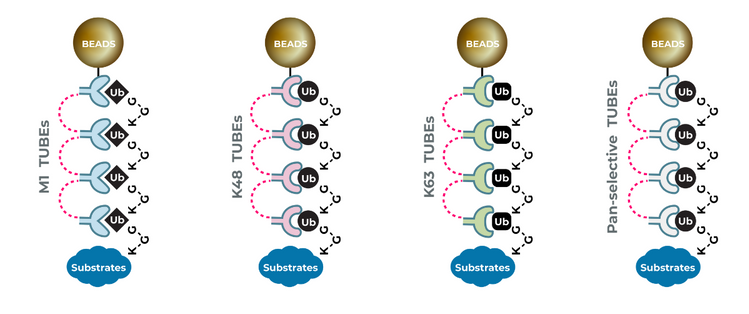
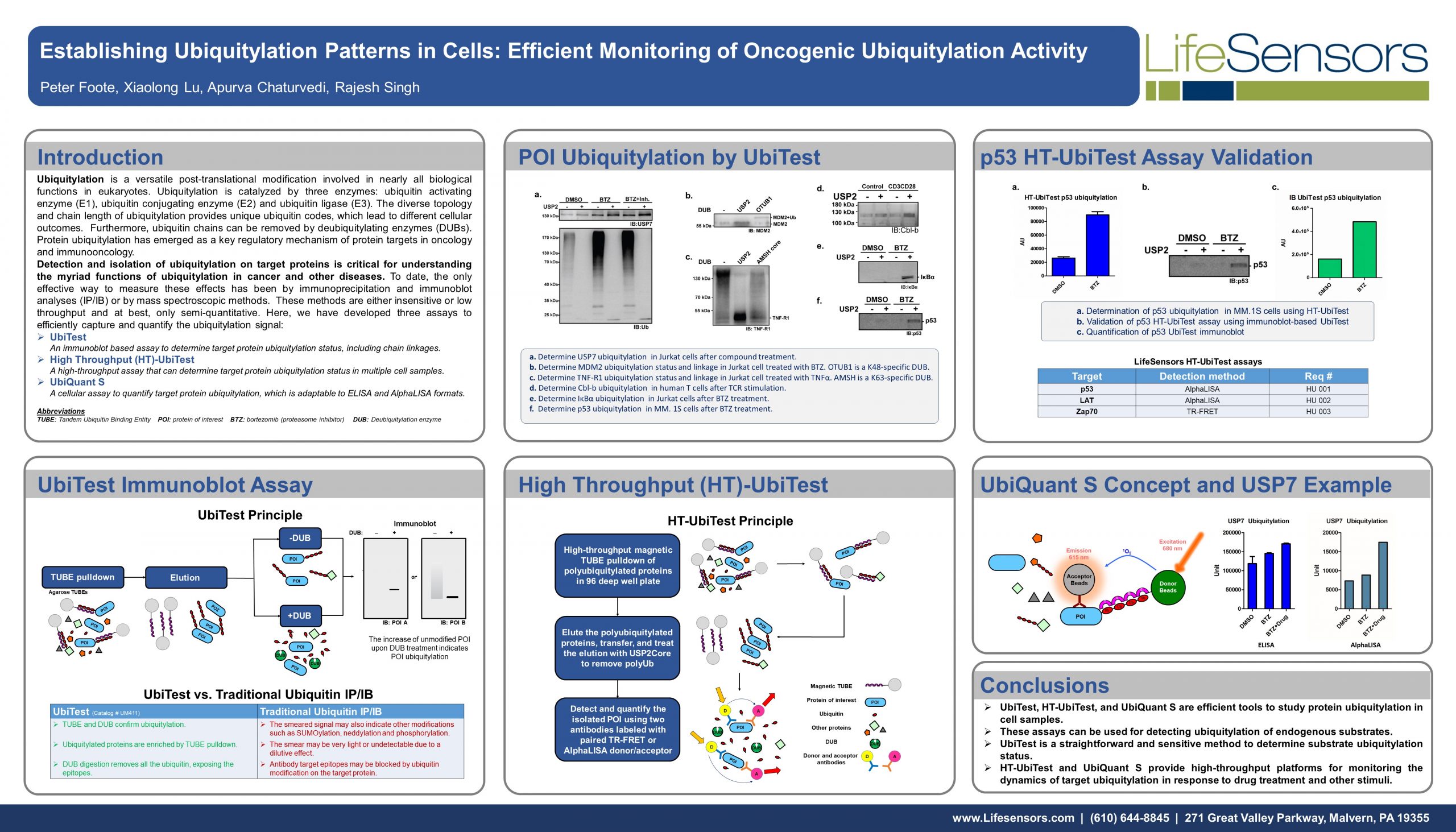
UbiTest: Detection of polyubiquitinated proteins using TUBE based screening platforms by Gauthami S. Jalagadugula The ubiquitin proteasome system (UPS) is a highly regulated mechanism that controls many cellular processes through…

Hijacking the natural mechanism of the Ubiquitin Proteasome System (UPS) is emerging as a powerful medium in drug discovery through which Dr. Kadimisetty uses “TUBEs” as ubiquitin-binding entities to monitor…

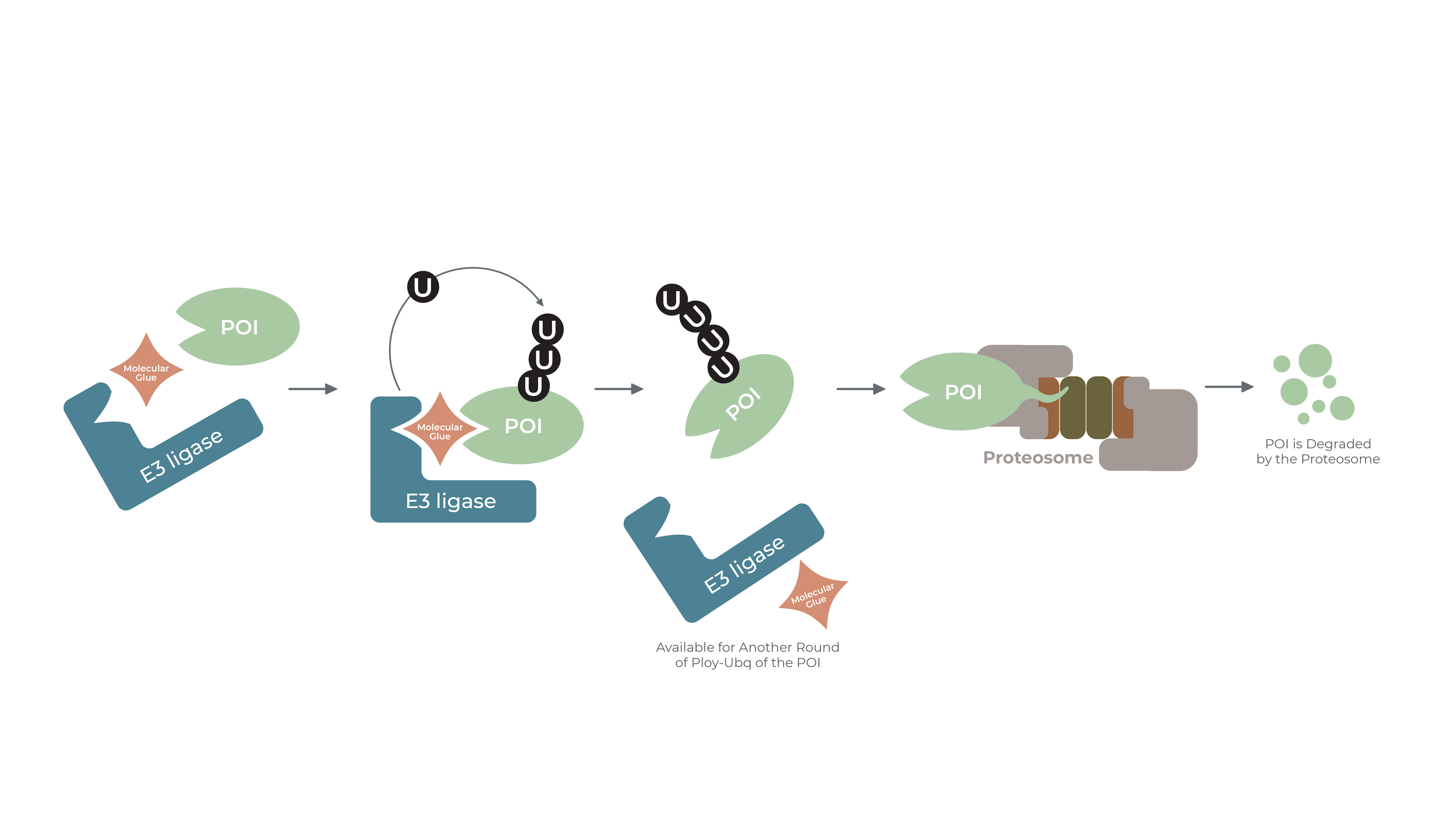
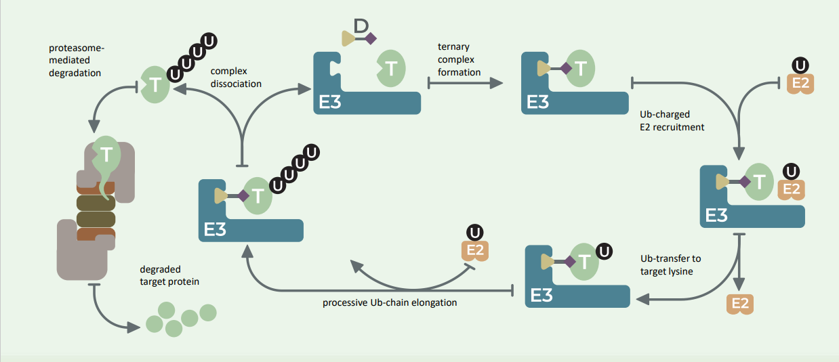
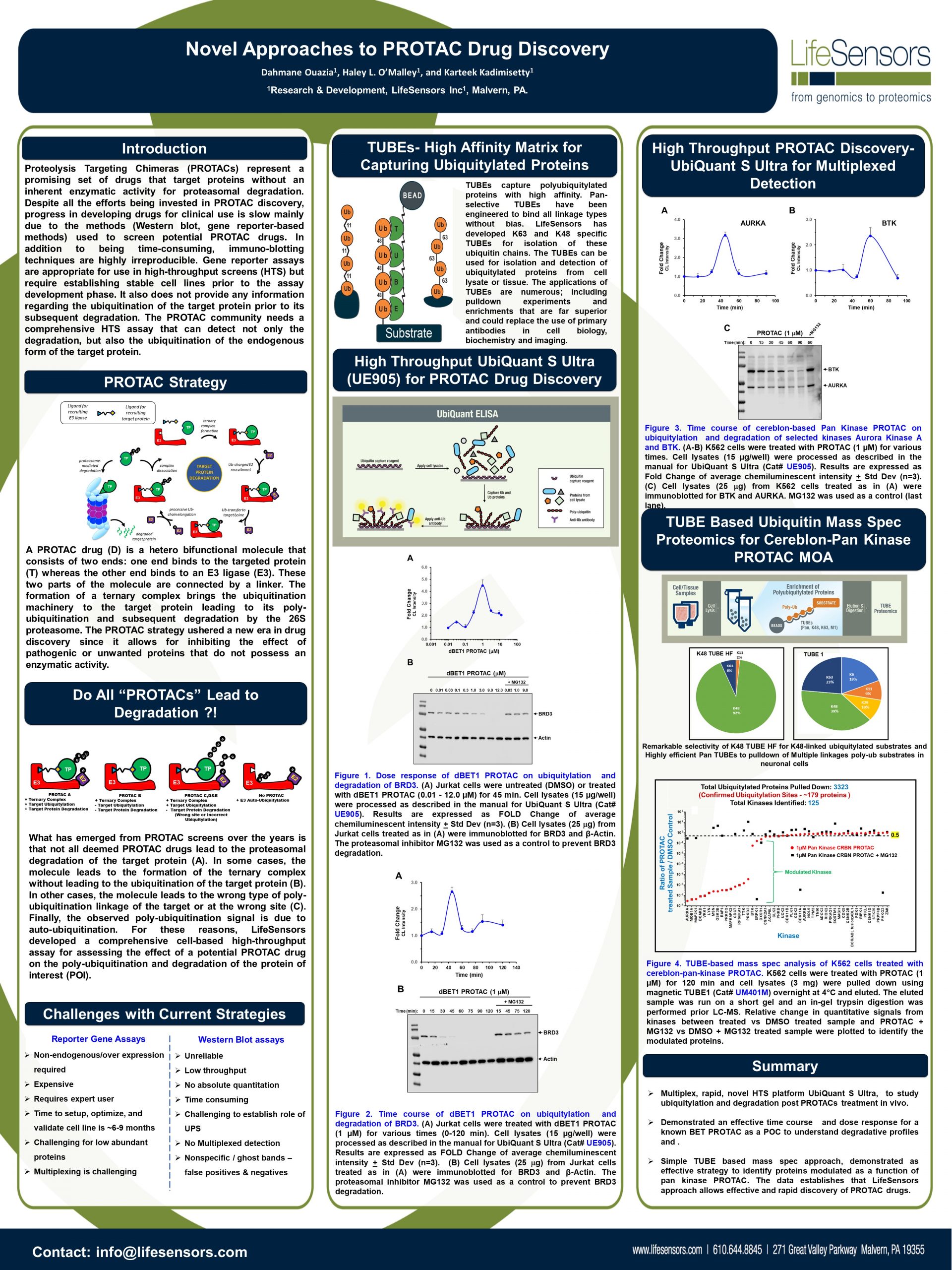
Discovering Unique Ubiquitin E3 Ligase Ligands to Unlock Targeted Protein degradation Gauthami Jalagadugula, Alexandra Russell1+, Kenneth Wallace1+, Janelle Pedroza and Abdul Haseeb1 * Introduction PROteolytic TArgeting Chimeras (PROTACs) have emerged…

PROTACs and Molecular Glues: High-Throughput Quantitative Assays for Rationale Design Authors: Janelle Pedroza1, Kenneth Wallace1, Derrick Padykula, Alexandra Russell1, and Karteek Kadimisetty*1 Introduction PROTACs have emerged as new class of…

MALVERN, Pa., SEP 1, 2022 – LifeSensors Inc., a Biotech and Drug Discovery Company focused on PROTAC, molecular glues, and neurodegeneration diagnostics, is pleased to announce that Lifesensors has published…



PROTAC Drug Discovery Webinar January 2022
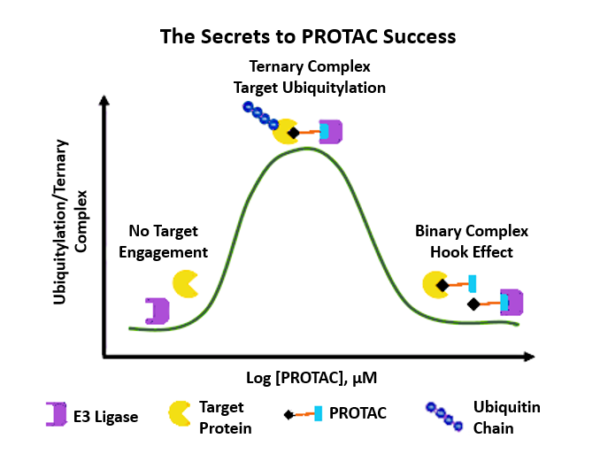
By: Rebekah Varghese, Derrick Padykula, & Jacob Huang All the FDA-approved drugs combined interact with only 2% of the proteins found in human cells, according to Dr. Brent Stockwell, a…
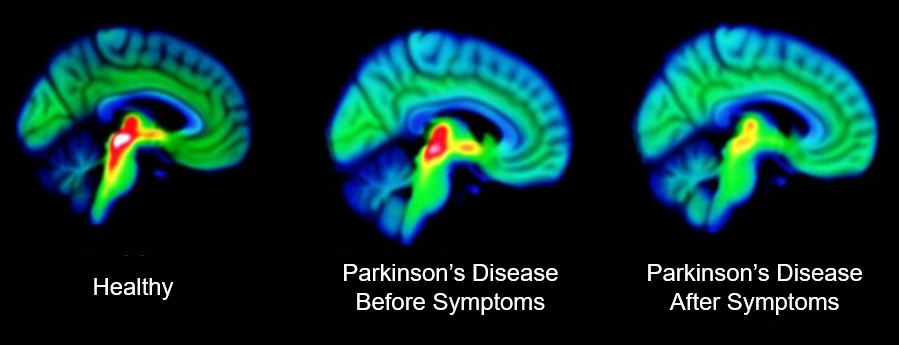
By: Rebekah Varghese and Derrick Padykula Parkinson’s Disease (PD) is the second most common neurodegenerative disease, affecting 1-2% of the population. Now, when you consider the world’s population to be…
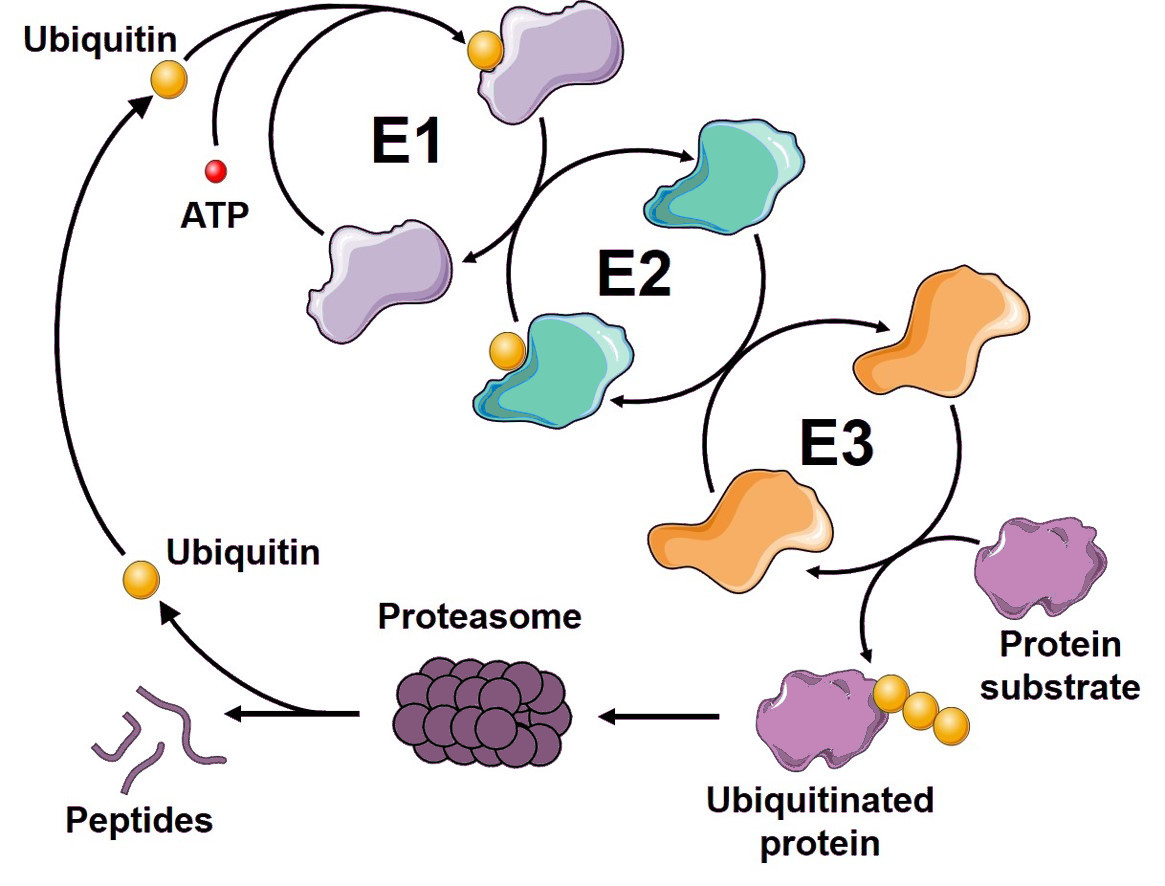
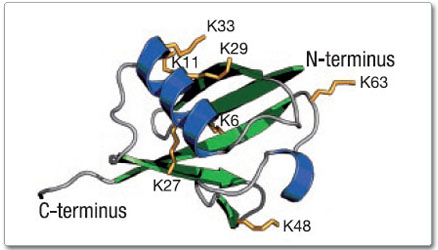
The Ubiquitin-Proteasome System: An Overview The Ubiquitin-Proteasome system (UPS) is a highly complex, temporally controlled and conserved pathway that plays a major role in a myriad on cellular functions….
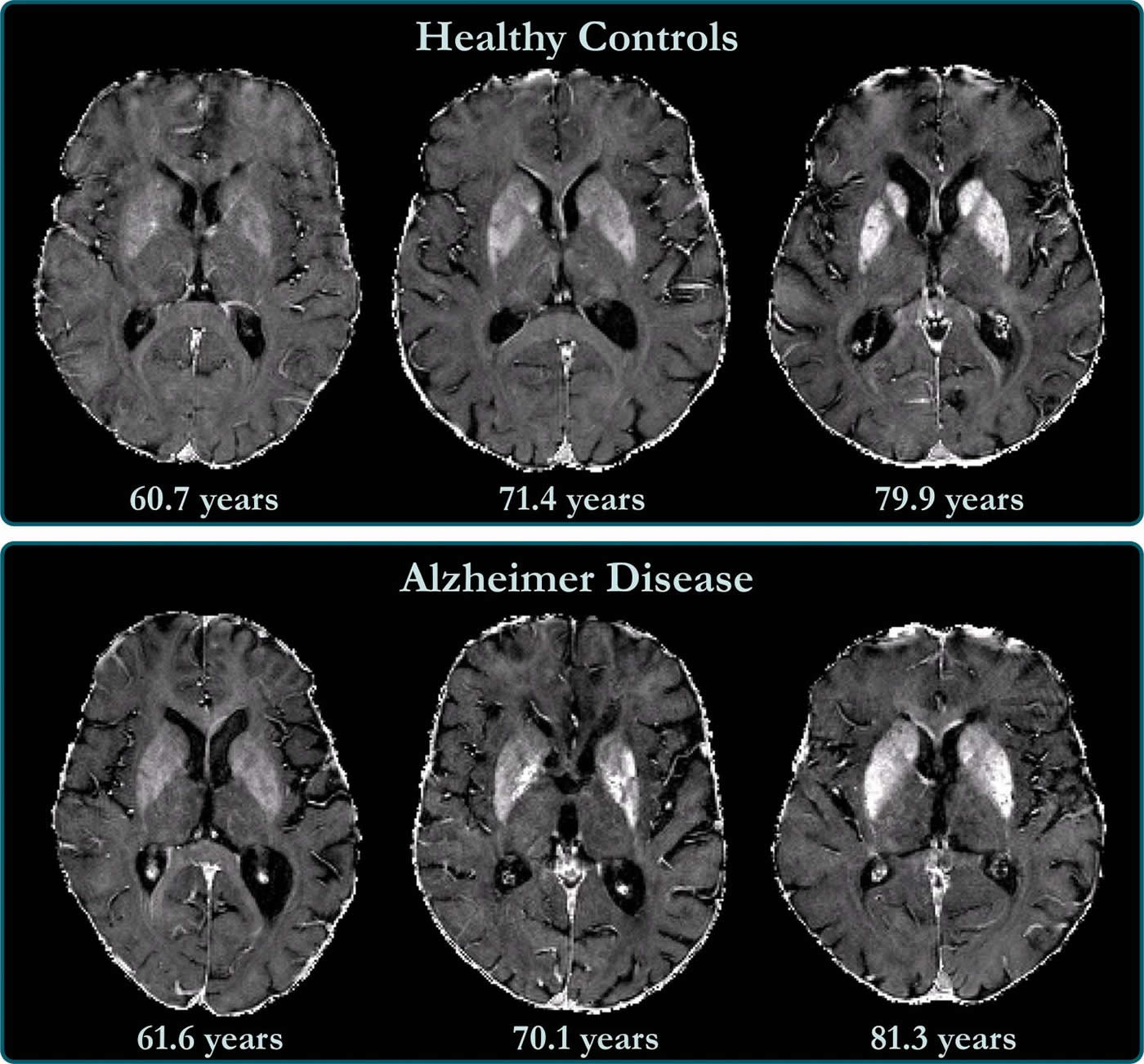
November is a month dedicated to the awareness of Alzheimer’s Disease, an age-related illness of the brain that causes cognitive and physiological decline. LifeSensors has great appreciation for our fellow…
DTE proteins, or ‘difficult-to-express’ proteins are a common hurdle for researchers in the lab. Due to difficulty with correct protein folding and general protein synthesis success, coronavirus proteins find themselves…

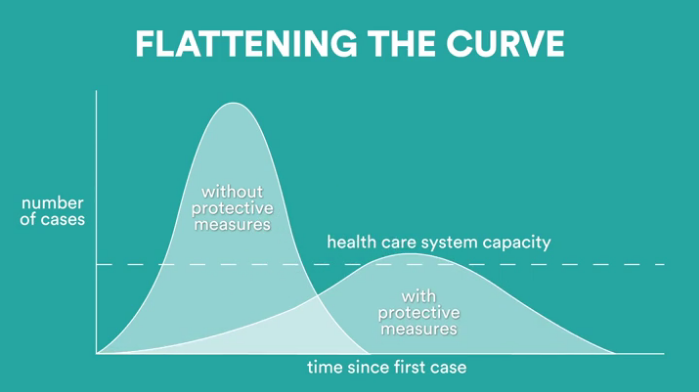
FDA Provisional Approval LifeSensors is proud to announce its provisional FDA approval for our COVID-19 IgG ELISA Detection Kit to address the critical need for COVID-19 diagnostics through serological testing….
IgG & Total Antibody Kit At LifeSensors, we value and believe that a reliable and affordable test against COVID-19 can protect patients and healthcare professionals from around the world by…
LifeSensors, Inc is pleased to announce it has been granted CLIA certification in light of the COVID-19 pandemic. With this new certification, LifeSensors will be able to aid with the…
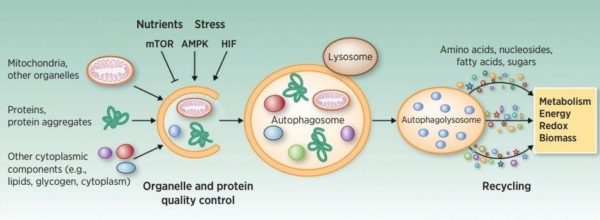
Autophagy Autophagy is a process by which eukaryotic cells degrade and recycle proteins and organelles using intracellular lysosomes. Malfunctions in autophagy have been shown to induce many diseases including cancer,…
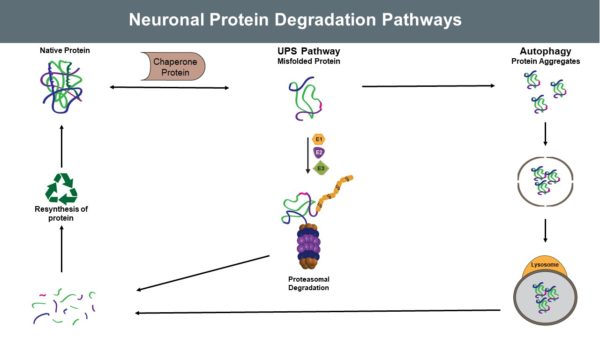
Ubiquitin and Neuroscience Neurodegenerative diseases are often characterized by excessive or dysfunctional proteins and protein aggregates, which induce specific pathological conditions. The ubiquitin proteasome system and autophagy play key roles…



Lewy Body Dementia (LBD) is a debilitating neurodegenerative disease that is increasingly becoming a public health concern. “In Lewy body dementia, alpha-synuclein forms into clumps inside neurons, starting in areas…

The Cold Spring Harbor Laboratory will be hosting a conference on the Ubiquitin Proteasome System (UPS) and associated Diseases April 23rd-27th. LifeSensors will be presenting on our most recent developments…

LifeSensors has spent years supporting the drug discovery efforts in Ubiquitin Proteasome System (UPS) research. We are excited to see this growing area of research gain more attention through conferences…

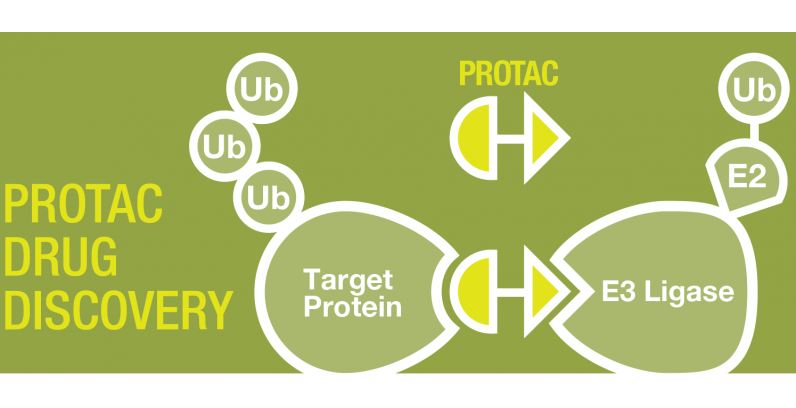

By: Mark Tingey, Katelyn Roberts, and Meredith Paterson Parkinson’s disease is often referred to as a silent disease. It often begins in very innocuous ways, small ticks and behaviors that…

LIFESENSORS TO SPONSOR DISCOVERY ON TARGET To help further scientific advancement in the field of ubiquitin, LifeSensors has partnered with CHI to assist with their Discovery on Target conference this…

UCHL1 The protein encoded by UCHL1 (Ubiquitin C-Terminal Hydrolase L1) is a thiol protease belonging to the C12 family of peptidases that hydrolyze peptide bonds at the C-terminal glycine of…
USP14 USP14 is a ubiquitin specific protease that associates with the proteasome. This association with the proteasome has been shown to enhance the enzymatic activity of the proteasome. Recent studies…
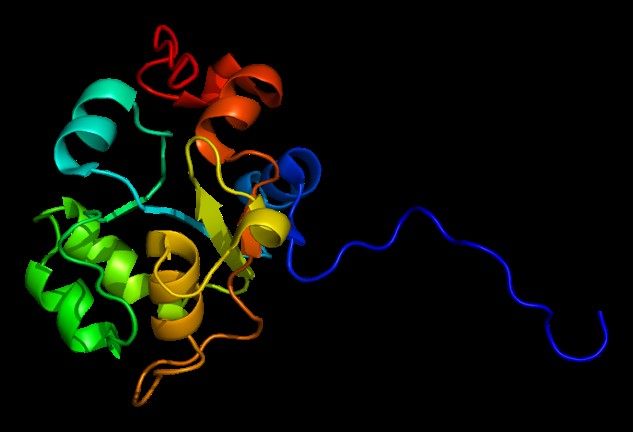
USP8 USP8 is associated with endosomal trafficking, it is not clear that this isopeptidase is involved in the commitment to lysosomal sorting. Additional research has shown that USP8 plays a…
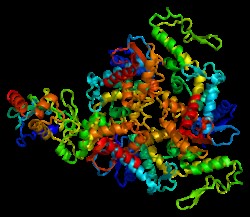
E6-AP E6-AP (E6-associated protein) is an E3 (ubiquitin protein ligase) that is involved in the conjugation of ubiquitin to target substrates along with E1 and E2 enzymes. It is a…
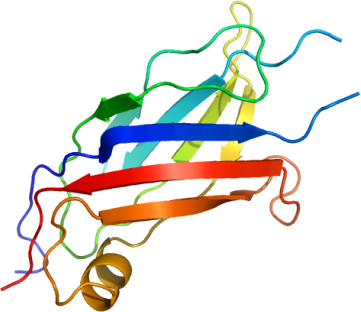
ITCH ITCH is a HECT E3, which influences immunity, the skin, and plays a role in cancer by targeting various transcription factors and growth factors. Additionally, ITCH functions by ubiquitinylating…
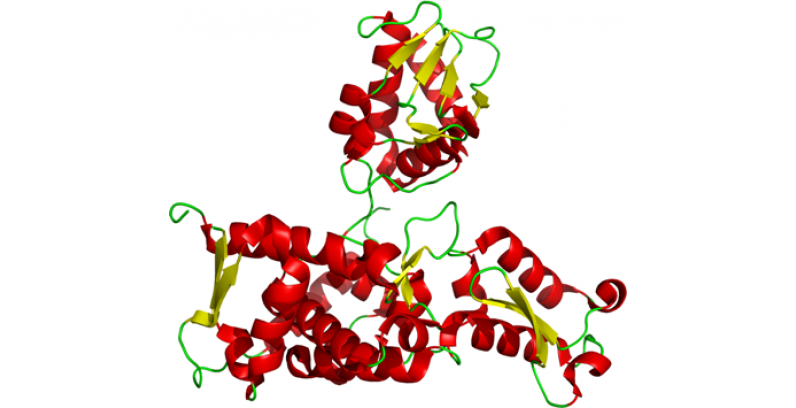
NEDD4 Nedd4 is an inner membrane bound HECT E3 that promotes endocytosis of sodium channels marked for degradation at FGFR1 receptors, with K63 linked ubiquitin selectivity. Nedd4 has also been…
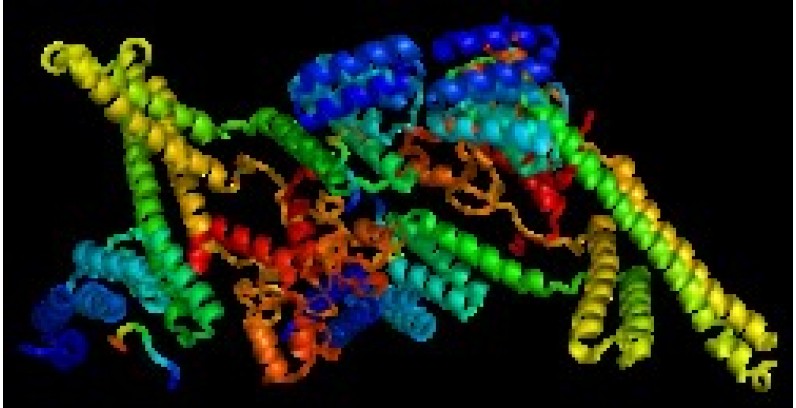
CHIP CHIP is an E3 that coordinates with Ataxin3 to mark misfolded chaperone proteins for UPS degradation. Deficiencies within this pathway are associated with rare disease, such as Spinocerebellar Ataxia,…
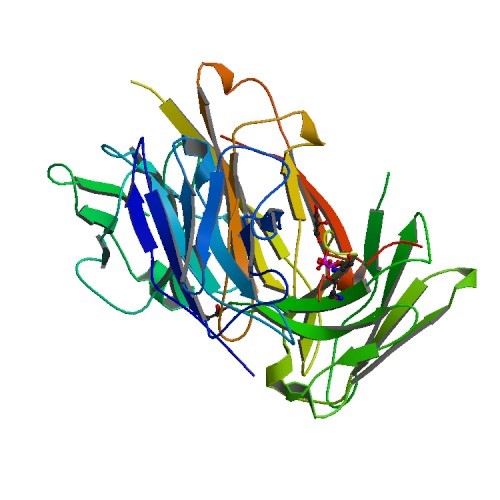
TAU PROTEINS PDB ID: 5D2V Within healthy neurons, tau proteins are responsible for stabilizing microtubules within the cell. Unfortunately, in instances associated with neurodegenerative disease these proteins are found to…
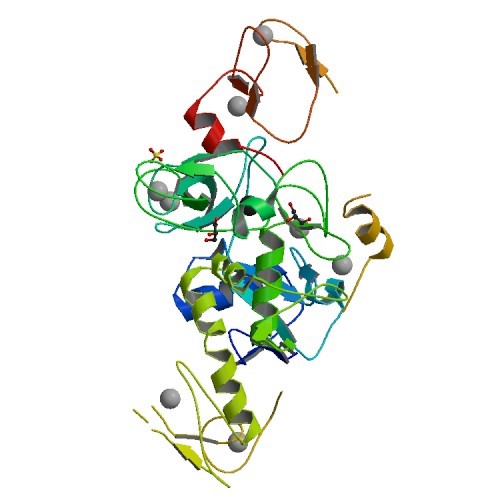
PARKIN PDB ID: 5C1Z Parkin is an E3 ligase, which ubiquitinylates proteins for degradation within neurons, and is primarily associated with mitophagy. By tagging damaged proteins with ubiquitin, the neuron…
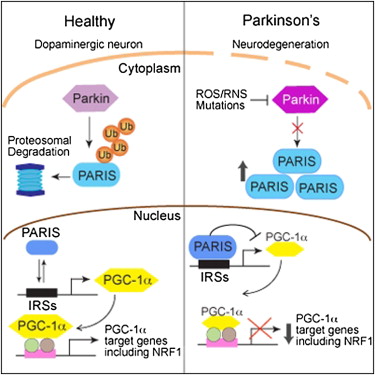
PARIS (ZNF746) Recently found to have drastic implications in Parkinson’s, researchers have shown PARIS to be a key regulating factor of neuronal death within the substantia nigra (the major site…
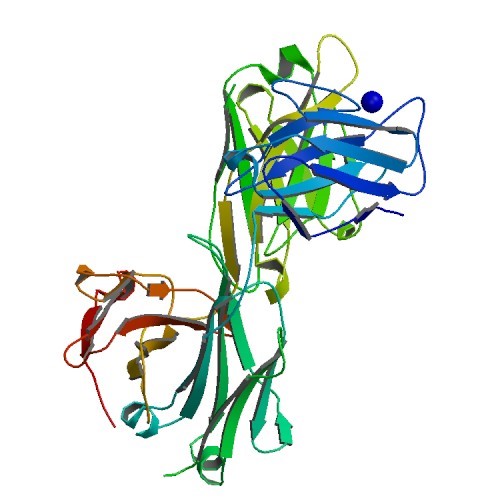
Β-AMYLOID PDB ID: 38KC Amyloid precursor proteins (APP), present in the fatty membrane of neuronal cells, undergo a series of enzymatic cleavages to create smaller proteins known as β-Amyloid. Sometimes,…
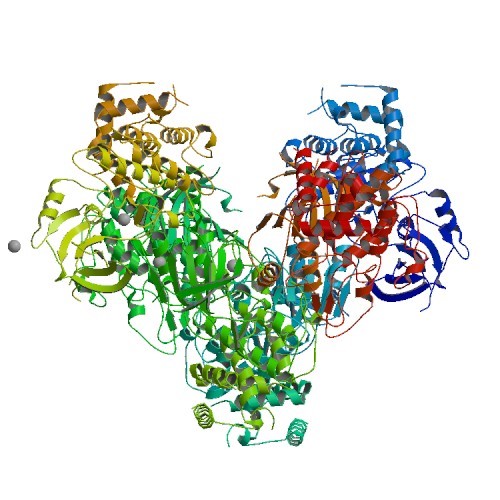
PINK1 PDB ID: 50AT PTEN induced putative kinase 1 (PINK1) is important in the role of mitophagy, once it accumulates on the outer membrane of a damaged mitochondria, it summons…
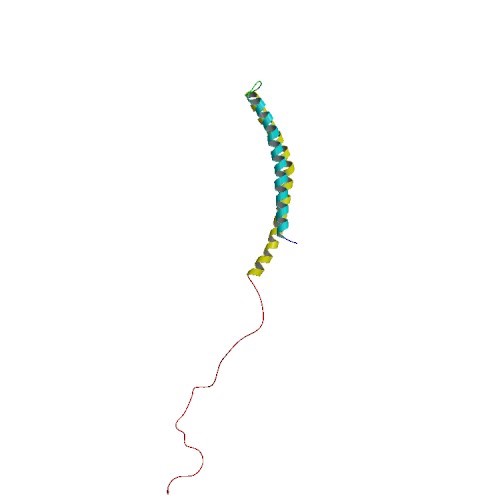
Α-SYNUCLEIN PDB ID: 1XQ8 α-Synuclein is mainly found in the brain and is present at the presynaptic terminal of neurons. Although its function in a healthy brain is currently unknown,…


PRESS RELEASE MAY 29, 2018 271 GREAT VALLEY PARKWAY MALVERN, PA 19355 LifeSensors Announces New Distribution Agreement with AmyJet in China LifeSensors Inc, a Biotech and Drug Discovery Company is…

LIFESENSORS IS PROUD TO SPONSOR THE 2018 FUSION UBIQUITIN CONFERENCE IN NASSAU, BAHAMAS It is with great pride that LifeSensors announces its sponsorship of the 2018 Fusion Conference The Ubiquitin…
Current Paradigms and Innovative Research Dear Colleagues, Alzheimer’s and Parkinson’s diseases are the silent killers. Despite a massive investment, the failure rate in drug development is 99.6%. The largest hurdle…
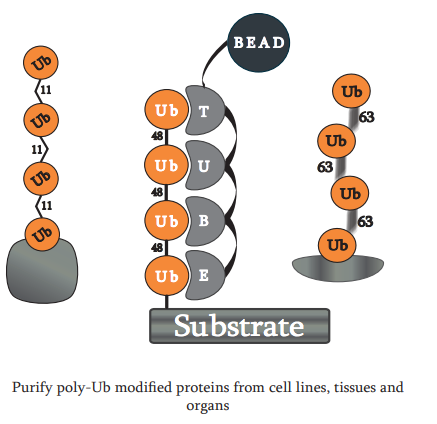
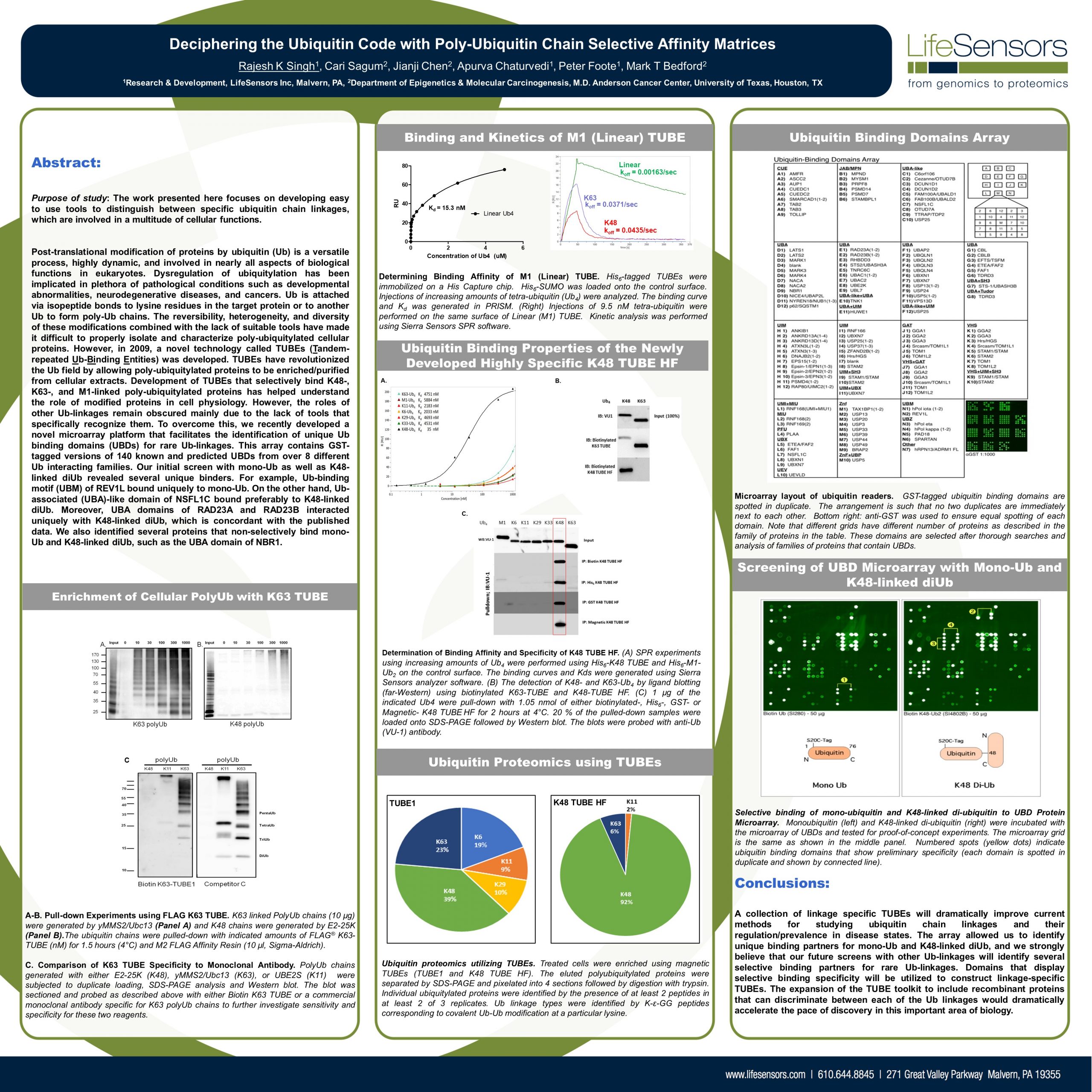
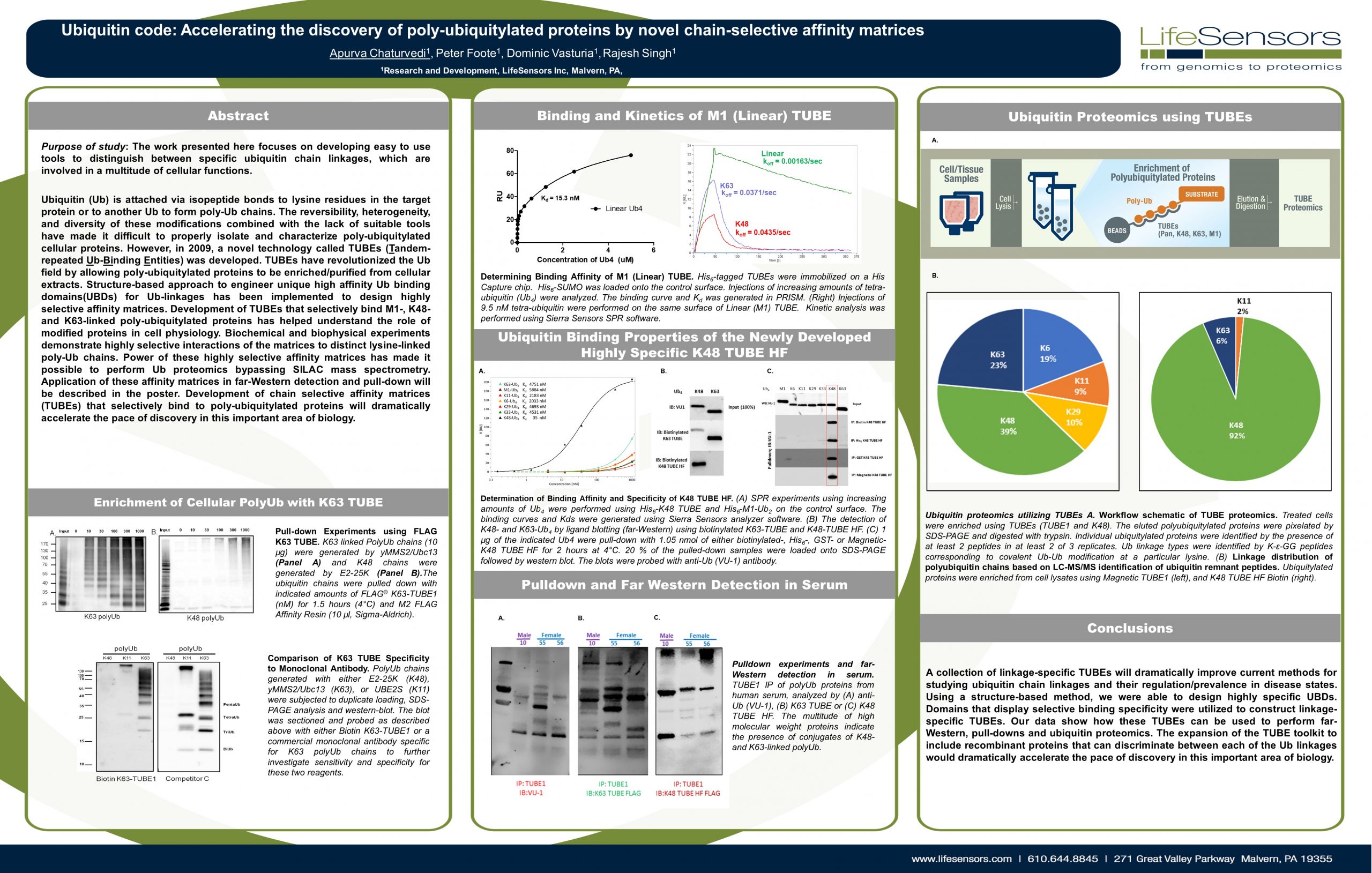
Dear Colleagues, Deciphering the Ubiquitin Code is the first step in utilizing the Ubiquitin Proteasome System (UPS) to drug the so-called undruggable targets. LifeSensors is proud to present a new…
UBIQUITIN: HOPE FOR THE FUTURE OF ALZHEIMER’S RESEARCH by: Mark Tingey and Peter Foote Uncle Walt stared at us, not recognizing us or remembering our names. His eyes, which were…
WE HAVE DEVELOPED A NEW WEBSITE FOR CUSTOMERS TO FOLLOW OUR INNOVATIONS AND THE FUTURE OF MEDICINE BY EXPLORING THE UBIQUITIN AND SUMO PATHWAY CHECK OUT OUR NEW FEATURES LIFESENSORS…
271 Great Valley Parkway
Malvern, PA 19355
Phone: 610.644.8845
Fax: 610.644.8616